- Services
- Application Migration Service for Toxicology Lab
Application Migration Service for Toxicology Lab
-
Blended Off-Shore & On-Shore Model
- Will bring in an on-site project manager with expertise in Pick Basic
- T & M contract but pay for only productive hours
-
Phased Implementation
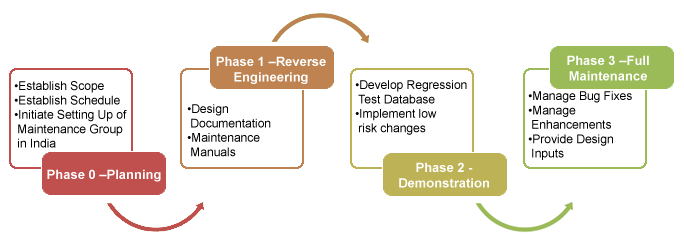
-
Manage Knowledge Transfer
- Ensure proper training procedures and testing to ensure that quality resources are working on the project
- Early low risk maintenance requests and expanding to maintaining and enhancing the full suite
- Keep about key existing resources intact to ensure continuity
|
|
The existing ERP system for a major toxicology laboratory was custom developed in the early nineties using an older client server platform. It was not meeting the company's current business requirements. The company could easily triple their business volume, but were limited by antiquated technology. Attempts to customize a popular ERP package had cost over 2 million dollars but had totally failed to meet user requirements. Eventually this package was abandoned and inteq was asked to provide a solution. The goal of this study was to analyze and assess the existing ERP system, assess the preparedness to respond to future changes in business such as supporting new market requirements for clinical testing and support of the needs of other laboratories.
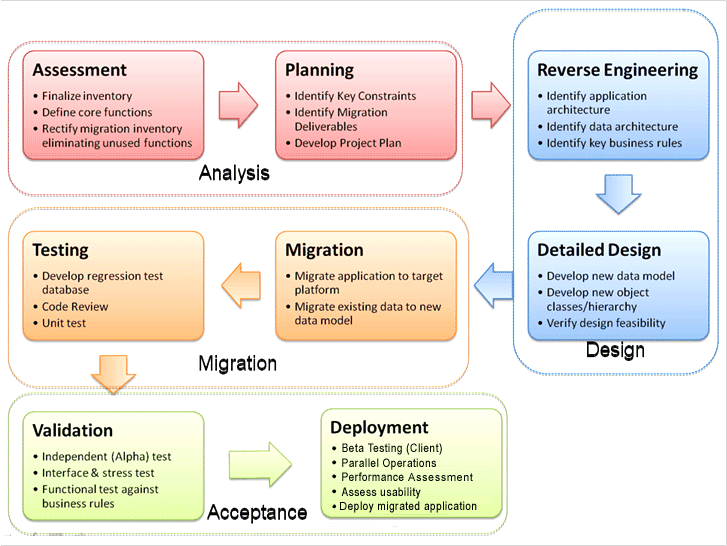
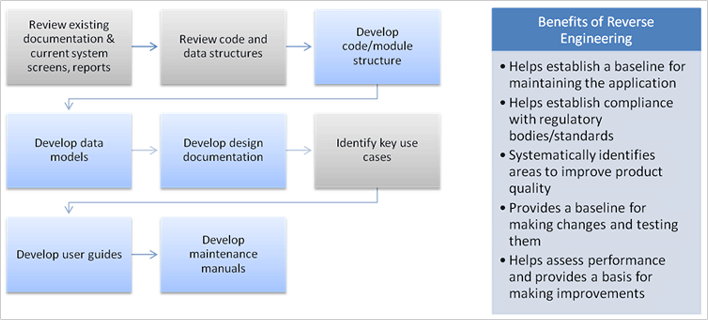
The specific areas studied in the assessment included the feasibility of a platform that is technologically current, easily maintainable and has the ability to easily grow and interface with a host of other customer information systems eliminating double entry, provide efficiencies in managing throughput, and better quality. Other objectives of the technology study was to enable management to have a real-time dashboard of performance, integrating financial, operational and customer information. The study recommended to include a data-warehouse that is seamlessly integrated with day-to-day operations to support data mining and trending.
To help implement the phases, inteq adopted its established on-shore/off-shore model. The on-shore work included developing the requirements and aligning it with the laboratory process. On-shore work was performed by inteq has strong understanding of the healthcare market and a solid expertise in HIPAA and other compliance regulations (21 CFR Part 11). inteq Corporation and technical analysts helped develop the RFP, the contract and quality acceptance criteria for the deliverables. They provided a fixed fee estimate and schedule. The critical aspect of the proposal was well articulated deliverables with quality acceptance criteria.
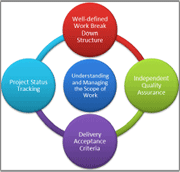
While the laboratory's CIO had complete management oversight, inteq Corporation maintained and tracked the deliveries from a cost, schedule, risk and quality perspective. The customer had the assurance of on-site management, cost efficient off-shore development and exchange moderators who oversees the service providers performance. The key success factors were: well defined requirements, well defined work-breakdown structure, project status tracking, independent quality assurance, and acceptance criteria tied to each deliverable.
inteq helped the laboratory save over 50% in total project costs using a blended off-shore/on-shore model. The laboratory has decided to use this blended global delivery model for other projects and has engaged inteq as their primary partner in designing, developing and maintaining their IT systems